Transmembrane Protein on:
[Wikipedia]
[Google]
[Amazon]
A transmembrane protein (TP) is a type of
integral membrane protein
An integral, or intrinsic, membrane protein (IMP) is a type of membrane protein that is permanently attached to the biological membrane. All ''transmembrane proteins'' are IMPs, but not all IMPs are transmembrane proteins. IMPs comprise a signi ...
that spans the entirety of the cell membrane
The cell membrane (also known as the plasma membrane (PM) or cytoplasmic membrane, and historically referred to as the plasmalemma) is a biological membrane that separates and protects the interior of all cells from the outside environment ( ...
. Many transmembrane proteins function as gateways to permit the transport of specific substances across the membrane. They frequently undergo significant conformational changes to move a substance through the membrane. They are usually highly hydrophobic
In chemistry, hydrophobicity is the physical property of a molecule that is seemingly repelled from a mass of water (known as a hydrophobe). In contrast, hydrophiles are attracted to water.
Hydrophobic molecules tend to be nonpolar and, t ...
and aggregate and precipitate in water. They require detergents or nonpolar solvents for extraction, although some of them (beta-barrel
In protein structures, a beta barrel is a beta sheet composed of tandem repeats that twists and coils to form a closed toroidal structure in which the first strand is bonded to the last strand (hydrogen bond). Beta-strands in many beta-barrels are ...
s) can be also extracted using denaturing agents.
The peptide sequence
Peptides (, ) are short chains of amino acids linked by peptide bonds. Long chains of amino acids are called proteins. Chains of fewer than twenty amino acids are called oligopeptides, and include dipeptides, tripeptides, and tetrapeptides.
A ...
that spans the membrane, or the transmembrane segment
A transmembrane domain (TMD) is a membrane-spanning protein domain. TMDs generally adopt an alpha helix topological conformation, although some TMDs such as those in porins can adopt a different conformation. Because the interior of the lipid bi ...
, is largely hydrophobic and can be visualized using the hydropathy plot Hydrophobicity scales are values that define the relative hydrophobicity or hydrophilicity of amino acid residues. The more positive the value, the more hydrophobic are the amino acids located in that region of the protein. These scales are commonly ...
. Depending on the number of transmembrane segments, transmembrane proteins can be classified as single-span (or bitopic) or multi-span (polytopic). Some other integral membrane proteins are called monotopic, meaning that they are also permanently attached to the membrane, but do not pass through it.
Types
Classification by structure
There are two basic types of transmembrane proteins:alpha-helical
The alpha helix (α-helix) is a common motif in the secondary structure of proteins and is a right hand-helix conformation in which every backbone N−H group hydrogen bonds to the backbone C=O group of the amino acid located four residues e ...
and beta barrel
In protein structures, a beta barrel is a beta sheet composed of tandem repeats that twists and coils to form a closed toroidal structure in which the first strand is bonded to the last strand (hydrogen bond). Beta-strands in many beta-barrels are ...
s. Alpha-helical proteins are present in the inner membranes of bacterial cells or the plasma membrane of eukaryotic cells, and sometimes in the bacterial outer membrane
The bacterial outer membrane is found in gram-negative bacteria. Its composition is distinct from that of the inner cytoplasmic cell membrane - among other things, the outer leaflet of the outer membrane of many gram-negative bacteria includes ...
. This is the major category of transmembrane proteins. In humans, 27% of all proteins have been estimated to be alpha-helical membrane proteins.
Beta-barrel proteins are so far found only in outer membranes of gram-negative bacteria
Gram-negative bacteria are bacteria that do not retain the crystal violet stain used in the Gram staining method of bacterial differentiation. They are characterized by their cell envelopes, which are composed of a thin peptidoglycan cell wall ...
, cell wall
A cell wall is a structural layer surrounding some types of cells, just outside the cell membrane. It can be tough, flexible, and sometimes rigid. It provides the cell with both structural support and protection, and also acts as a filtering mech ...
s of gram-positive bacteria
In bacteriology, gram-positive bacteria are bacteria that give a positive result in the Gram stain test, which is traditionally used to quickly classify bacteria into two broad categories according to their type of cell wall.
Gram-positive bact ...
, outer membranes of mitochondria
A mitochondrion (; ) is an organelle found in the Cell (biology), cells of most Eukaryotes, such as animals, plants and Fungus, fungi. Mitochondria have a double lipid bilayer, membrane structure and use aerobic respiration to generate adenosi ...
and chloroplasts
A chloroplast () is a type of membrane-bound organelle known as a plastid that conducts photosynthesis mostly in plant cell, plant and algae, algal cells. The photosynthetic pigment chlorophyll captures the energy from sunlight, converts it, ...
, or can be secreted as pore-forming toxin
Pore-forming proteins (PFTs, also known as pore-forming toxins) are usually produced by bacteria, and include a number of protein exotoxins but may also be produced by other organisms such as apple snails that produce perivitellin-2 or earthwo ...
s. All beta-barrel transmembrane proteins have simplest up-and-down topology, which may reflect their common evolutionary origin and similar folding mechanism.
In addition to the protein domains, there are unusual transmembrane elements formed by peptides. A typical example is gramicidin A
Gramicidin, also called gramicidin D, is a mix of ionophoric antibiotics, gramicidin A, B and C, which make up about 80%, 5%, and 15% of the mix, respectively. Each has 2 isoforms, so the mix has 6 different types of gramicidin molecules. They c ...
, a peptide that forms a dimeric transmembrane β-helix. This peptide is secreted by gram-positive bacteria
In bacteriology, gram-positive bacteria are bacteria that give a positive result in the Gram stain test, which is traditionally used to quickly classify bacteria into two broad categories according to their type of cell wall.
Gram-positive bact ...
as an antibiotic
An antibiotic is a type of antimicrobial substance active against bacteria. It is the most important type of antibacterial agent for fighting bacterial infections, and antibiotic medications are widely used in the treatment and prevention of ...
. A transmembrane polyproline-II helix has not been reported in natural proteins. Nonetheless, this structure was experimentally observed in specifically designed artificial peptides.
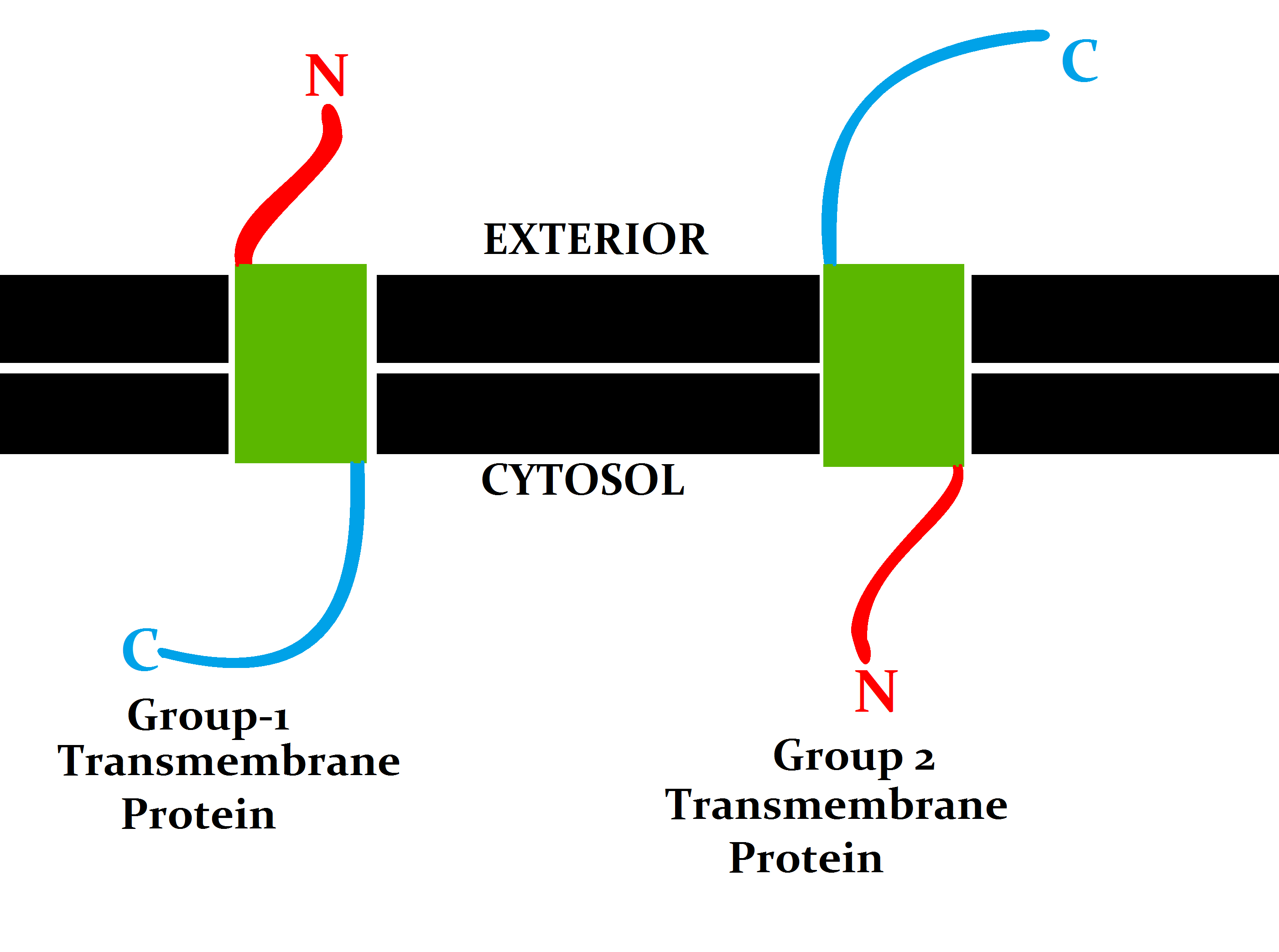
Classification by topology
This classification refers to the position of the protein N- and C-termini on the different sides of thelipid bilayer
The lipid bilayer (or phospholipid bilayer) is a thin polar membrane made of two layers of lipid molecules. These membranes are flat sheets that form a continuous barrier around all cells. The cell membranes of almost all organisms and many vir ...
. Types I, II, III and IV are single-pass molecules. Type I transmembrane proteins are anchored to the lipid membrane with a stop-transfer anchor sequence and have their N-terminal domains targeted to the endoplasmic reticulum (ER) lumen during synthesis (and the extracellular space, if mature forms are located on cell membranes). Type II and III are anchored with a signal-anchor sequence, with type II being targeted to the ER lumen with its C-terminal domain, while type III have their N-terminal domains targeted to the ER lumen. Type IV is subdivided into IV-A, with their N-terminal domains targeted to the cytosol and IV-B, with an N-terminal domain targeted to the lumen. The implications for the division in the four types are especially manifest at the time of translocation and ER-bound translation, when the protein has to be passed through the ER membrane in a direction dependent on the type.

3D structure
Membrane protein
Membrane proteins are common proteins that are part of, or interact with, biological membranes. Membrane proteins fall into several broad categories depending on their location. Integral membrane proteins are a permanent part of a cell membrane ...
structures can be determined by X-ray crystallography
X-ray crystallography is the experimental science determining the atomic and molecular structure of a crystal, in which the crystalline structure causes a beam of incident X-rays to diffract into many specific directions. By measuring the angles ...
, electron microscopy or NMR spectroscopy
Nuclear magnetic resonance spectroscopy, most commonly known as NMR spectroscopy or magnetic resonance spectroscopy (MRS), is a spectroscopic technique to observe local magnetic fields around atomic nuclei. The sample is placed in a magnetic fiel ...
. The most common tertiary structures
Biomolecular structure is the intricate folded, three-dimensional shape that is formed by a molecule of protein, DNA, or RNA, and that is important to its function. The structure of these molecules may be considered at any of several length s ...
of these proteins are transmembrane helix bundle and beta barrel
In protein structures, a beta barrel is a beta sheet composed of tandem repeats that twists and coils to form a closed toroidal structure in which the first strand is bonded to the last strand (hydrogen bond). Beta-strands in many beta-barrels are ...
. The portion of the membrane proteins that are attached to the lipid bilayer (see annular lipid shell Annular lipids (also called shell lipids or boundary lipids) are a set of lipids or lipidic molecules which preferentially bind or stick to the surface of membrane proteins in biological cells. They constitute a layer, or an annulus/ shell, of lipid ...
) consist mostly of hydrophobic amino acids.White, Stephen. "General Principle of Membrane Protein Folding and Stability". Stephen White Laboratory Homepage. 10 Nov. 2009. web.
Membrane proteins which have hydrophobic surfaces, are relatively flexible and are expressed at relatively low levels. This creates difficulties in obtaining enough protein and then growing crystals. Hence, despite the significant functional importance of membrane proteins, determining atomic resolution structures for these proteins is more difficult than globular proteins. As of January 2013 less than 0.1% of protein structures determined were membrane proteins despite being 20–30% of the total proteome. Due to this difficulty and the importance of this class of proteins methods of protein structure prediction based on hydropathy plots, the positive inside rule and other methods have been developed.
Thermodynamic stability and folding
Stability of alpha-helical transmembrane proteins
Transmembrane
A transmembrane protein (TP) is a type of integral membrane protein that spans the entirety of the cell membrane. Many transmembrane proteins function as gateways to permit the transport of specific substances across the membrane. They frequentl ...
alpha-helical
The alpha helix (α-helix) is a common motif in the secondary structure of proteins and is a right hand-helix conformation in which every backbone N−H group hydrogen bonds to the backbone C=O group of the amino acid located four residues e ...
(α-helical) proteins are unusually stable judging from thermal denaturation studies, because they do not unfold completely within the membranes (the complete unfolding would require breaking down too many α-helical H-bond
In chemistry, a hydrogen bond (or H-bond) is a primarily electrostatic force of attraction between a hydrogen (H) atom which is covalently bound to a more electronegative "donor" atom or group (Dn), and another electronegative atom bearing a l ...
s in the nonpolar media). On the other hand, these proteins easily ''misfold'', due to non-native aggregation in membranes, transition to the molten globule
In molecular biology, the term molten globule (MG) refers to protein states that are more or less compact (hence the "globule"), but are lacking the specific tight packing of amino acid residues which creates the solid state-like tertiary structu ...
states, formation of non-native disulfide bonds
In biochemistry, a disulfide (or disulphide in British English) refers to a functional group with the structure . The linkage is also called an SS-bond or sometimes a disulfide bridge and is usually derived by the coupling of two thiol groups. In ...
, or unfolding of peripheral regions and nonregular loops that are locally less stable.
It is also important to properly define the ''unfolded state
In biochemistry, denaturation is a process in which proteins or nucleic acids lose the quaternary structure, tertiary structure, and secondary structure which is present in their native state, by application of some external stress or compou ...
''. The ''unfolded state'' of membrane proteins in detergent micelles
A micelle () or micella () (plural micelles or micellae, respectively) is an aggregate (or supramolecular assembly) of surfactant amphipathic lipid molecules dispersed in a liquid, forming a colloidal suspension (also known as associated coll ...
is different from that in the thermal denaturation experiments. This state represents a combination of folded hydrophobic α-helices and partially unfolded segments covered by the detergent. For example, the "unfolded" bacteriorhodopsin
Bacteriorhodopsin is a protein used by Archaea, most notably by haloarchaea, a class of the Euryarchaeota. It acts as a proton pump; that is, it captures light energy and uses it to move protons across the membrane out of the cell. The resulting ...
in SDS micelles has four transmembrane α-helices folded, while the rest of the protein is situated at the micelle-water interface and can adopt different types of non-native amphiphilic
An amphiphile (from the Greek αμφις amphis, both, and φιλíα philia, love, friendship), or amphipath, is a chemical compound possessing both hydrophilic (''water-loving'', polar) and lipophilic (''fat-loving'') properties. Such a compo ...
structures. Free energy differences between such detergent-denatured and native states are similar to stabilities of water-soluble proteins (< 10 kcal/mol).
Folding of α-helical transmembrane proteins
Refolding of α-helical transmembrane proteins ''in vitro'' is technically difficult. There are relatively few examples of the successful refolding experiments, as forbacteriorhodopsin
Bacteriorhodopsin is a protein used by Archaea, most notably by haloarchaea, a class of the Euryarchaeota. It acts as a proton pump; that is, it captures light energy and uses it to move protons across the membrane out of the cell. The resulting ...
. ''In vivo'', all such proteins are normally folded co-translationally within the large transmembrane translocon The translocon (also known as a translocator or translocation channel) is a complex of proteins associated with the translocation of polypeptides across membranes. In eukaryotes the term translocon most commonly refers to the complex that transport ...
. The translocon channel provides a highly heterogeneous environment for the nascent transmembrane α-helices. A relatively polar amphiphilic α-helix can adopt a transmembrane orientation in the translocon (although it would be at the membrane surface or unfolded ''in vitro''), because its polar residues can face the central water-filled channel of the translocon. Such mechanism is necessary for incorporation of polar α-helices into structures of transmembrane proteins. The amphiphilic helices remain attached to the translocon until the protein is completely synthesized and folded. If the protein remains unfolded and attached to the translocon for too long, it is degraded by specific "quality control" cellular systems.
Stability and folding of beta-barrel transmembrane proteins
Stability of beta barrel (β-barrel) transmembrane proteins is similar to stability of water-soluble proteins, based on chemical denaturation studies. Some of them are very stable even in chaotropic agents and high temperature. Their folding ''in vivo'' is facilitated by water-soluble chaperones, such as protein Skp. It is thought that β-barrel membrane proteins come from one ancestor even having different number of sheets which could be added or doubled during evolution. Some studies show a huge sequence conservation among different organisms and also conserved amino acids which hold the structure and help with folding.3D structures
Light absorption-driven transporters
*Bacteriorhodopsin
Bacteriorhodopsin is a protein used by Archaea, most notably by haloarchaea, a class of the Euryarchaeota. It acts as a proton pump; that is, it captures light energy and uses it to move protons across the membrane out of the cell. The resulting ...
-like proteins including rhodopsin (see also opsin
Animal opsins are G-protein-coupled receptors and a group of proteins made light-sensitive via a chromophore, typically retinal. When bound to retinal, opsins become Retinylidene proteins, but are usually still called opsins regardless. Most pro ...
)
*Bacterial photosynthetic reaction centre
A photosynthetic reaction center is a complex of several proteins, pigments and other co-factors that together execute the primary energy conversion reactions of photosynthesis. Molecular excitations, either originating directly from sunlight or t ...
s and photosystem
Photosystems are functional and structural units of protein complexes involved in photosynthesis. Together they carry out the primary photochemistry of photosynthesis: the absorption of light and the transfer of energy and electrons. Photosystems ...
s I and II
*Light-harvesting complex
A light-harvesting complex consists of a number of chromophores which are complex subunit proteins that may be part of a larger super complex of a photosystem, the functional unit in photosynthesis. It is used by plants and photosynthetic bacteri ...
es from bacteria
Bacteria (; singular: bacterium) are ubiquitous, mostly free-living organisms often consisting of one biological cell. They constitute a large domain of prokaryotic microorganisms. Typically a few micrometres in length, bacteria were among ...
and chloroplasts
A chloroplast () is a type of membrane-bound organelle known as a plastid that conducts photosynthesis mostly in plant cell, plant and algae, algal cells. The photosynthetic pigment chlorophyll captures the energy from sunlight, converts it, ...
Oxidoreduction-driven transporters
*Transmembrane cytochrome b-like proteins:coenzyme Q - cytochrome c reductase
A cofactor is a non-protein chemical compound or metallic ion that is required for an enzyme's role as a catalyst (a catalyst is a substance that increases the rate of a chemical reaction). Cofactors can be considered "helper molecules" that ass ...
(cytochrome bc1 ); cytochrome b6f complex
The cytochrome ''b''6''f'' complex (plastoquinol—plastocyanin reductase; ) is an enzyme found in the thylakoid membrane in chloroplasts of plants, cyanobacteria, and green algae, that catalyzes the transfer of electrons from plastoquinol to ...
; formate dehydrogenase, respiratory nitrate reductase; succinate - coenzyme Q reductase
Succinate dehydrogenase (SDH) or succinate-coenzyme Q reductase (SQR) or respiratory complex II is an enzyme complex, found in many bacterial cells and in the inner mitochondrial membrane of eukaryotes. It is the only enzyme that participates in ...
(fumarate reductase); and succinate dehydrogenase
Succinate dehydrogenase (SDH) or succinate-coenzyme Q reductase (SQR) or respiratory complex II is an enzyme complex, found in many bacterial cells and in the inner mitochondrial membrane of eukaryotes. It is the only enzyme that participates i ...
. See electron transport chain
An electron transport chain (ETC) is a series of protein complexes and other molecules that transfer electrons from electron donors to electron acceptors via redox reactions (both reduction and oxidation occurring simultaneously) and couples th ...
.
*Cytochrome c oxidase
The enzyme cytochrome c oxidase or Complex IV, (was , now reclassified as a translocasEC 7.1.1.9 is a large transmembrane protein complex found in bacteria, archaea, and mitochondria of eukaryotes.
It is the last enzyme in the respiratory elect ...
s from bacteria
Bacteria (; singular: bacterium) are ubiquitous, mostly free-living organisms often consisting of one biological cell. They constitute a large domain of prokaryotic microorganisms. Typically a few micrometres in length, bacteria were among ...
and mitochondria
A mitochondrion (; ) is an organelle found in the Cell (biology), cells of most Eukaryotes, such as animals, plants and Fungus, fungi. Mitochondria have a double lipid bilayer, membrane structure and use aerobic respiration to generate adenosi ...
Electrochemical potential-driven transporters
*Proton or sodium translocating F-type and V-typeATPase
ATPases (, Adenosine 5'-TriPhosphatase, adenylpyrophosphatase, ATP monophosphatase, triphosphatase, SV40 T-antigen, ATP hydrolase, complex V (mitochondrial electron transport), (Ca2+ + Mg2+)-ATPase, HCO3−-ATPase, adenosine triphosphatase) are ...
s
P-P-bond hydrolysis-driven transporters
*P-typecalcium ATPase
Ca2+ ATPase is a form of P-ATPase that transfers calcium after a muscle has contracted. The two kinds of calcium ATPase are:
*Plasma membrane Ca2+ ATPase (PMCA)
*Sarcoplasmic reticulum Ca2+ ATPase (SERCA)
Plasma membrane Ca2+ ATPase (PMCA)
P ...
(five different conformations)
*Calcium ATPase regulators phospholamban
Phospholamban, also known as PLN or PLB, is a micropeptide protein that in humans is encoded by the ''PLN'' gene. Phospholamban is a 52-amino acid integral membrane protein that regulates the calcium (Ca2+) pump in cardiac muscle cells.
Funct ...
and sarcolipin
Sarcolipin is a micropeptide protein that in humans is encoded by the ''SLN'' gene.
Function
Sarcoplasmic reticulum Ca2+-ATPases are transmembrane proteins that catalyze the ATP-dependent transport of Ca2+ from the cytosol into the lumen of t ...
*ABC transporter
The ATP-binding cassette transporters (ABC transporters) are a transport system superfamily that is one of the largest and possibly one of the oldest gene families. It is represented in all extant phyla, from prokaryotes to humans. ABC transpo ...
s
*General secretory pathway 440px
Secretion is the movement of material from one point to another, such as a secreted chemical substance from a cell or gland. In contrast, excretion is the removal of certain substances or waste products from a cell or organism. The classica ...
(Sec) translocon The translocon (also known as a translocator or translocation channel) is a complex of proteins associated with the translocation of polypeptides across membranes. In eukaryotes the term translocon most commonly refers to the complex that transport ...
(preprotein translocase SecY)
Porters (uniporters, symporters, antiporters)
* Mitochondrialcarrier protein
A membrane transport protein (or simply transporter) is a membrane protein involved in the movement of ions, small molecules, and macromolecules, such as another protein, across a biological membrane. Transport proteins are integral transmembrane ...
s
*Major Facilitator Superfamily (Glycerol-3-phosphate transporter, Lactose permease The permeases are membrane transport proteins, a class of multipass transmembrane proteins that allow the diffusion of a specific molecule in or out of the cell in the direction of a concentration gradient, a form of facilitated diffusion.
The per ...
, and Multidrug transporter EmrD)
* Resistance-nodulation-cell division (multidrug efflux transporter AcrB, see multidrug resistance
Multiple drug resistance (MDR), multidrug resistance or multiresistance is antimicrobial resistance shown by a species of microorganism to at least one antimicrobial drug in three or more antimicrobial categories. Antimicrobial categories are c ...
)
*Dicarboxylate/amino acid:cation symporter (proton glutamate symporter)
*Monovalent cation/proton antiporter (Sodium/proton antiporter 1 NhaA)
*Neurotransmitter
A neurotransmitter is a signaling molecule secreted by a neuron to affect another cell across a synapse. The cell receiving the signal, any main body part or target cell, may be another neuron, but could also be a gland or muscle cell.
Neuro ...
sodium symporter
*Ammonia transporters
*Drug/Metabolite Transporter (small multidrug resistance transporter EmrE - the structures are retracted as erroneous)
Alpha-helical channels including ion channels
*Voltage-gated ion channel
Voltage-gated ion channels are a class of transmembrane proteins that form ion channels that are activated by changes in the electrical membrane potential near the channel. The membrane potential alters the conformation of the channel proteins, ...
like, including potassium channel
Potassium channels are the most widely distributed type of ion channel found in virtually all organisms. They form potassium-selective pores that span cell membranes. Potassium channels are found in most cell types and control a wide variety of cel ...
s KcsA and KvAP, and inward-rectifier potassium ion channel Kirbac
* Large-conductance mechanosensitive channel, MscL
* Small-conductance mechanosensitive ion channel (MscS)
* CorA metal ion transporters
*Ligand-gated ion channel
Ligand-gated ion channels (LICs, LGIC), also commonly referred to as ionotropic receptors, are a group of transmembrane ion-channel proteins which open to allow ions such as Na+, K+, Ca2+, and/or Cl− to pass through the membrane in res ...
of neurotransmitter
A neurotransmitter is a signaling molecule secreted by a neuron to affect another cell across a synapse. The cell receiving the signal, any main body part or target cell, may be another neuron, but could also be a gland or muscle cell.
Neuro ...
receptors (acetylcholine receptor
An acetylcholine receptor (abbreviated AChR) is an integral membrane protein that responds to the binding of acetylcholine, a neurotransmitter.
Classification
Like other transmembrane receptors, acetylcholine receptors are classified according ...
)
*Aquaporin
Aquaporins, also called water channels, are channel proteins from a larger family of major intrinsic proteins that form pores in the membrane of biological cells, mainly facilitating transport of water between cells. The cell membranes of a v ...
s
*Chloride channel
Chloride channels are a superfamily of poorly understood ion channels specific for chloride. These channels may conduct many different ions, but are named for chloride because its concentration ''in vivo'' is much higher than other anions. Severa ...
s
*Outer membrane auxiliary proteins (polysaccharide transporter) - α-helical transmembrane proteins from the outer bacterial membrane
Enzymes
*Methane monooxygenase
Methane monooxygenase (MMO) is an enzyme capable of oxidizing the C-H bond in methane as well as other alkanes. Methane monooxygenase belongs to the class of oxidoreductase enzymes ().
There are two forms of MMO: the well-studied soluble form (s ...
*Rhomboid protease
The rhomboid proteases are a family of enzymes that exist in almost all species. They are proteases: they cut the polypeptide chain of other proteins. This proteolytic cleavage is irreversible in cells, and an important type of cellular regulation ...
*Disulfide bond
In biochemistry, a disulfide (or disulphide in British English) refers to a functional group with the structure . The linkage is also called an SS-bond or sometimes a disulfide bridge and is usually derived by the coupling of two thiol groups. In ...
formation protein (DsbA-DsbB complex)
Proteins with alpha-helical transmembrane anchors
*T cell receptor
The T-cell receptor (TCR) is a protein complex found on the surface of T cells, or T lymphocytes, that is responsible for recognizing fragments of antigen as peptides bound to major histocompatibility complex (MHC) molecules. The binding b ...
transmembrane dimerization domain ]
*Cytochrome c nitrite reductase complex
*Steryl-sulfate sulfohydrolase
*Stannin
*Glycophorin
A glycophorin is a sialoglycoprotein of the membrane of a red blood cell. It is a membrane-spanning protein and carries sugar molecules. It is heavily glycosylated (60%). Glycophorins are rich in sialic acid, which gives the red blood cells a ver ...
A dimer
*Inovirus (filamentous phage
Filamentous bacteriophage is a family of viruses (''Inoviridae'') that infect bacteria. The phages are named for their filamentous shape, a worm-like chain (long, thin and flexible, reminiscent of a length of cooked spaghetti), about 6 nm ...
) major coat protein
*Pilin
Pilin refers to a class of fibrous proteins that are found in pilus structures in bacteria. These structures can be used for the exchange of genetic material, or as a cell adhesion mechanism. Although not all bacteria have pili or fimbriae, ba ...
*Pulmonary surfactant
Pulmonary surfactant is a surface-active complex of phospholipids and proteins formed by type II alveolar cells. The proteins and lipids that make up the surfactant have both hydrophilic and hydrophobic
In chemistry, hydrophobicity is t ...
-associated protein
* Monoamine oxidases A and B
*Fatty acid amide hydrolase
Fatty acid amide hydrolase or FAAH (, oleamide hydrolase, anandamide amidohydrolase) is a member of the serine hydrolase family of enzymes. It was first shown to break down anandamide in 1993. In humans, it is encoded by the gene ''FAAH''.;
Fun ...
*Cytochrome P450 oxidase
Cytochromes P450 (CYPs) are a superfamily of enzymes containing heme as a cofactor that functions as monooxygenases. In mammals, these proteins oxidize steroids, fatty acids, and xenobiotics, and are important for the clearance of various compo ...
s
* Corticosteroid 11β-dehydrogenases .
*Signal Peptide Peptidase
In molecular biology, the Signal Peptide Peptidase (SPP) is a type of protein that specifically cleaves parts of other proteins. It is an intramembrane aspartyl protease with the conserved active site motifs 'YD' and 'GxGD' in adjacent transmemb ...
*Membrane protease specific for a stomatin homolog
Beta-barrels composed of a single polypeptide chain
*Beta barrels from eight beta-strands and with "shear number" of ten (''n=8, S=10''). They include: ** OmpA-like transmembrane domain (OmpA) ** Virulence-related outer membrane protein family (OmpX) ** Outer membrane protein W family (OmpW) ** Antimicrobial peptide resistance and lipid A acylation protein family (PagP) ** Lipid A deacylase PagL ** Opacity family porins (NspA) *Autotransporter domain
In molecular biology, an autotransporter domain is a structural domain found in some bacterial outer membrane proteins. The domain is always located at the C-terminal end of the protein and forms a beta-barrel structure. The barrel is oriented in ...
(''n=12,S=14'')
* FadL outer membrane protein transport family, including Fatty acid
In chemistry, particularly in biochemistry, a fatty acid is a carboxylic acid with an aliphatic chain, which is either saturated or unsaturated. Most naturally occurring fatty acids have an unbranched chain of an even number of carbon atoms, fr ...
transporter FadL (''n=14,S=14'')
* General bacterial porin family
General bacterial porins are a family of porin proteins from the outer membranes of Gram-negative bacteria. The porins act as molecular filters for hydrophilic compounds. They are responsible for the 'molecular sieve' properties of the outer mem ...
, known as trimeric porins
Porins are beta barrel proteins that cross a cellular membrane and act as a pore, through which molecules can diffuse. Unlike other membrane transport proteins, porins are large enough to allow passive diffusion, i.e., they act as channels tha ...
(''n=16,S=20'')
* Maltoporin, or sugar porins
Porins are beta barrel proteins that cross a cellular membrane and act as a pore, through which molecules can diffuse. Unlike other membrane transport proteins, porins are large enough to allow passive diffusion, i.e., they act as channels tha ...
(''n=18,S=22'')
* Nucleoside-specific porin (''n=12,S=16'')
* Outer membrane phospholipase A1(''n=12,S=16'')
*TonB-dependent receptors
Outer membrane receptors, also known as TonB-dependent receptors, are a family of beta barrel proteins named for their localization in the outer membrane of gram-negative bacteria. TonB complexes sense signals from the outside of bacterial cells ...
and their plug domain. They are ligand-gated outer membrane channels (''n=22,S=24''), including cobalamin
Vitamin B12, also known as cobalamin, is a water-soluble vitamin involved in metabolism. It is one of eight B vitamins. It is required by animals, which use it as a cofactor in DNA synthesis, in both fatty acid and amino acid metabolism. It ...
transporter BtuB, Fe(III)-pyochelin receptor FptA, receptor FepA, ferric hydroxamate uptake receptor FhuA, transporter FecA, and pyoverdine receptor FpvA
* Outer membrane protein OpcA family (''n=10,S=12'') that includes outer membrane protease
A protease (also called a peptidase, proteinase, or proteolytic enzyme) is an enzyme that catalyzes (increases reaction rate or "speeds up") proteolysis, breaking down proteins into smaller polypeptides or single amino acids, and spurring the ...
OmpT and adhesin/invasin OpcA protein
*Outer membrane protein G
Outer membrane protein G (OmpG) is a porin, a channel proteins in the outer membrane of Gram-negative bacteria.
''Escherichia coli'' OmpG forms a 14-stranded beta-barrel and in contrast to most porins, appears to function as a monomer. The cen ...
porin family (''n=14,S=16'')
Note: ''n'' and ''S'' are, respectively, the number of beta-strands and the "shear number" of the beta-barrel
In protein structures, a beta barrel is a beta sheet composed of tandem repeats that twists and coils to form a closed toroidal structure in which the first strand is bonded to the last strand (hydrogen bond). Beta-strands in many beta-barrels are ...
Beta-barrels composed of several polypeptide chains
* Trimeric autotransporter (''n=12,S=12'') * Outer membrane efflux proteins, also known as trimeric outer membrane factors (n=12,S=18) including TolC and multidrug resistance proteins *MspA porin
Mycobacterial porins are a group of transmembrane beta-barrel proteins produced by mycobacteria, which allow hydrophilic nutrients to enter the bacterium.Membrane protein
Membrane proteins are common proteins that are part of, or interact with, biological membranes. Membrane proteins fall into several broad categories depending on their location. Integral membrane proteins are a permanent part of a cell membrane ...
* Membrane topology
Topology of a transmembrane protein refers to locations of N- and C-termini of membrane-spanning polypeptide chain with respect to the inner or outer sides of the biological membrane occupied by the protein.
Several databases provide experimenta ...
* Transmembrane domain
A transmembrane domain (TMD) is a membrane-spanning protein domain. TMDs generally adopt an alpha helix topological conformation, although some TMDs such as those in porins can adopt a different conformation. Because the interior of the lipid bil ...
* Transmembrane receptor
Cell surface receptors (membrane receptors, transmembrane receptors) are receptor (biochemistry), receptors that are embedded in the cell membrane, plasma membrane of cell (biology), cells. They act in cell signaling by receiving (binding to) ex ...
s
References
{{DEFAULTSORT:Transmembrane Protein Integral membrane proteins